MPMED-SEZ is East Africa's only Special Economic Zone encoding both pharmaceutical manufacturing and medical device assembly within a single governed industrial site — addressing a $491M annual domestic market (MSD) with 80%+ import dependency, and access to 331M+ (EAC) and 450M+ (SADC) regional consumers.
Tanzania imports over 80% of its medicines — with only 19 domestic pharmaceutical manufacturing facilities vs 726 authorized foreign manufacturers. MPMED-SEZ is the designated site for expanding domestic production of generic medicines, biologics, APIs, vaccines, and traditional medicines at WHO-GMP certified industrial scale.
Over 90% of Tanzania's medical devices and 99% of diagnostics are imported — 157,444 import permits issued between 2022–2025 alone. MPMED-SEZ creates East Africa's first dedicated medical device assembly zone — precision surgical devices, clinical consumables, IVD diagnostics, and digital health hardware.
Authoritative regulatory data from TMDA — WHO Maturity Level 3 (first in Africa), ISO 9001:2015 certified, with WHO-prequalified laboratories — showing the scale of Tanzania's healthcare manufacturing gap.
Medical Stores Department (MSD) — Tanzania's national public health supply chain agency — provides structured market assurance for local manufacturers through centralised procurement, serving 8,800+ health facilities across 10 zones.
PIAT is a government-established, high-level coordination mechanism to facilitate, de-risk, and fast-track pharmaceutical manufacturing investment in Tanzania — composed of senior decision-makers from across the health, investment, and industrial sectors. Investors are assessed through a transparent, standardised due diligence framework across 11 weighted areas.
Pharmaceutical manufacturing is a top government priority under President H.E. Dr. Samia Suluhu Hassan, with strong leadership backing.
Multiple government agencies working together to streamline processes and provide unified investor support.
Integrated with national health, investment, and industrial development agendas for coherent support.
Clear directive to PIAT to move from policy discussion to actionable projects and tangible results.
PIAT is composed of senior decision-makers ensuring approvals are taken at the highest level — fast-tracking approvals, avoiding administrative delays, and bringing together diverse stakeholders to drive Tanzania's pharmaceutical industry forward.
PIAT works across three pillars to create the most enabling environment for pharmaceutical manufacturers entering Tanzania — de-risking entry, streamlining approvals, and aligning incentives with infrastructure.
PIAT mitigates risks and barriers through the Green-Lane Approval System — priority regulatory approvals, accelerated decision-making, coordinated government support, and connecting investors with local partners including MSD Medipharm.
Green-Lane Approval System — coordinating across government agencies to simplify and expedite approvals. Rolling, accelerated review of EOIs with approval granted promptly to investors demonstrating technical capacity, financial strength, and strategic alignment.
Aligning tax benefits and fiscal incentives with development of industrial parks, reliable utilities, and supporting infrastructure for pharmaceutical operations.
Manufacturing and supplying essential pharmaceuticals — antibiotics (22.4% of 8,465 registered medicines), cardiovascular agents (12.4%), analgesics (13.1%), GI medicines (16.6%). MSD provides 15% price preference and dedicated tenders. $491M annual procurement market.
Zero human biologics or vaccine manufacturing plants in Tanzania (TMDA) — only 3 veterinary vaccine facilities exist. A critical gap for investing in vaccine and biologic therapy production to strengthen public health infrastructure. EAC/SADC regional market reach.
Expanding local manufacturing of critical medical supplies — 157,444 import permits issued 2022–2025 for devices and diagnostics. Top imports: condoms, sutures, syringes, gloves, IV sets, reagents, blood bags. Reduce 90%+ import dependence.
Neither API nor excipients manufacturing facilities exist in Tanzania or the entire EAC region — 100% of APIs are imported (MSD/TMDA). Developing medium-to-long-term capability to produce Active Pharmaceutical Ingredients and other critical inputs. Addresses the deepest supply chain vulnerability.
From initial expression of interest to full-scale manufacturing establishment — PIAT guides you through every step with coordinated government support.
Investors express their interest in partnering with PIAT and investing in Tanzania's pharmaceutical manufacturing sector through the MPMED-SEZ portal or direct engagement with the facilitation desk.
PIAT conducts a preliminary due diligence assessment across 11 weighted areas using a standardised scoring tool (1–5 scale). Investors scoring 85–100 are Highly Recommended; 70–84 Recommended; 55–69 Conditional. Assessment based on business plan, feasibility study, interviews, and site visits.
PIAT collaborates with the investor to develop a customised localisation roadmap — outlining milestones for technology transfer, skills development, and local value addition within the SEZ framework.
PIAT provides streamlined support facilitating the regulatory approval process and addressing operational requirements — TMDA, TISEZA, BRELA coordination for smooth establishment within MPMED-SEZ.
Set up the manufacturing facility in Tanzania, leveraging PIAT's support and MPMED-SEZ infrastructure, and gradually scale up production to meet local and regional demand across EAC/SADC markets.
PIAT uses a standardised Preliminary Due Diligence Assessment Tool across 11 weighted areas. Score 1–5 on each criterion, with an overall weighted score determining your recommendation level. No hidden criteria — here's exactly what we evaluate.
Sector-specific resources for pharmaceutical and medical device investors — zone data, investment profiles, regulatory guides, PIAT demand forecasts, and feasibility tools published by the Ministry of Health.
Boundary maps, pharmaceutical cluster, medical device cluster, logistics zone, R&D park, infrastructure status, and the complete 600ha master development plan.
Detailed profiles for generic medicines, biologics, API synthesis, vaccines, and traditional medicines — market demand (8,465 registered products, 900–1,000 new applications/year), IRR benchmarks, GMP requirements, and TMDA 60-day pathway.
Profiles for precision devices, clinical consumables, IVDs, and digital health hardware — 1,906 registered products, 1,000–1,500 new applications/year, ISO 13485 requirements, and TMDA device registration guide.
Tanzania's SEZ investment regulations under TISEZA (Act No. 6 of 2025), reduced corporate tax, zero import duty, 100% foreign ownership rules, profit repatriation guarantees, and preferential procurement policy.
MoH-approved financial models for pharmaceutical manufacturing plants, medical device assembly facilities, GMP warehousing, cold-chain logistics, and IVD production units.
Ministry-convened forums for pharmaceutical and medical device investors — PIAT sector briefings, TISEZA site visits, TMDA pre-licensing consultations, and business matching with MoH facilitation.
Priority pharmaceutical and medical device investment opportunities within MPMED-SEZ — identified by the Ministry of Health, PIAT, and the Tanzania Investment Centre. Essential medicines and vaccines are PIAT strategic focus areas.
WHO-GMP facility for Tanzania's essential medicines list — solid dosage, injectables, oral liquids, topicals. Only 19 domestic manufacturers vs 726 foreign; antibiotics 22.4% of registered medicines. TMDA 60-day fast-track. MSD 15% price preference and dedicated local tenders.
High-volume production of gloves, syringes, IV sets, wound care, PPE, and surgical disposables. 157,444 import permits issued 2022–2025 — Tanzania's largest import substitution opportunity by unit volume. Direct MoH public procurement contracts available.
Active Pharmaceutical Ingredient production — no API or excipients manufacturing facilities exist in Tanzania or the entire EAC region; 100% of APIs are imported (TMDA/MSD). Chemical engineering infrastructure, dedicated API cluster. Zero domestic or regional competition.
Reagent manufacturing, rapid test kit assembly, and laboratory diagnostic equipment for malaria, HIV, TB, and NCD panels. 99% of diagnostics are imported (TMDA). WHO Emergency Use Listing pathway supported within the zone.
Purpose-built GMP-compliant cold-chain storage and distribution for vaccines, biologics, and temperature-sensitive medicines — serving MPMED-SEZ manufacturers and the wider East African supply chain.
Clinical research, biomedical innovation, and health technology development — linked to Muhimbili National Hospital and Tanzanian universities. Academic and private sector partnership-ready facility within the zone.
Tanzania's TISEZA investment framework (Act No. 6 of 2025) delivers one of East Africa's most competitive incentive environments for pharmaceutical and medical device manufacturers.
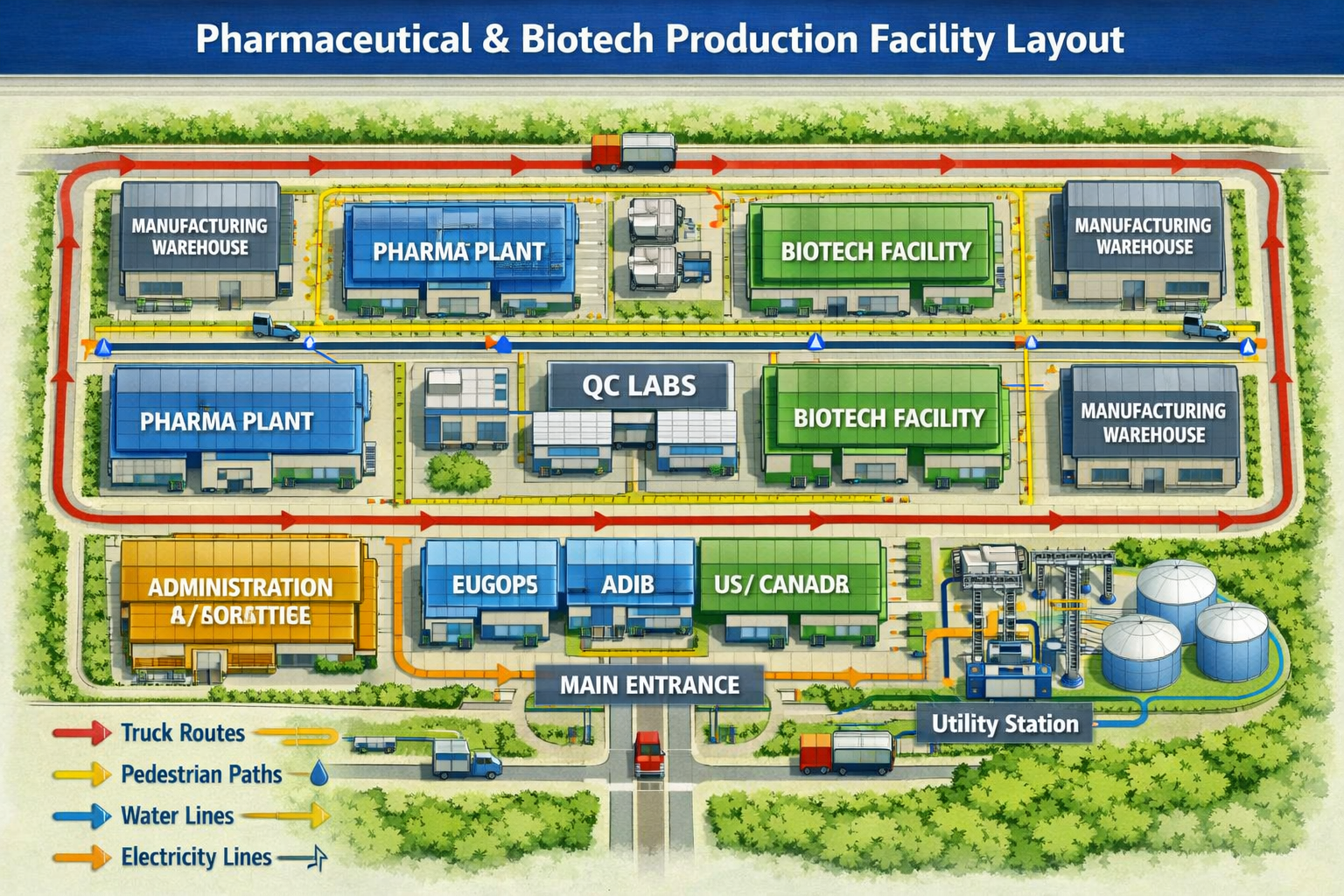
Mloganzila Plateau, 25km west of Dar es Salaam — the zone is physically designed around two industrial clusters with shared logistics, utilities, and regulatory infrastructure.
Dedicated pharmaceutical zone — GMP-compliant factory shells, pharmaceutical-grade water, controlled environments, API production area.
ISO 13485 clean-room zone — device assembly units, sterilisation services, sterile packaging, TMDA device registration support office.
GMP-compliant temperature-controlled warehousing and distribution connecting both manufacturing clusters to domestic and East African markets.
Clinical research, biomedical engineering, and health technology development — linked to Muhimbili National Hospital campus.
Official policy documents, pharmaceutical manufacturing guides, medical device regulatory frameworks, feasibility templates, and market intelligence — published by the Ministry of Health.
MPMED-SEZ's regulatory framework includes separate licensing tracks for pharmaceutical manufacturers and medical device companies — both coordinated through PIAT and TISEZA's One Stop Facilitation Centre.
Step-by-step from pre-application consultation to manufacturing authorisation — accelerated 60-day timeline for domestic products. Reduced registration fees (USD 400 vs USD 2,000 for imports).
Specific to medical device and IVD assembly within MPMED-SEZ — ISO 13485 certification aligned with TMDA device registration requirements.
Complete all eight sections as required by the Ministry of Health EOI Call. Fields marked * are mandatory. Submissions are reviewed on a rolling, accelerated basis under the Green-Lane Approval System.